Q-approximation of operating characteristics of clinical trial designs
S. Gentile, D. E. Schwartz, R. Saha, L. Trippa
•
Mar 20, 2026
Abstract
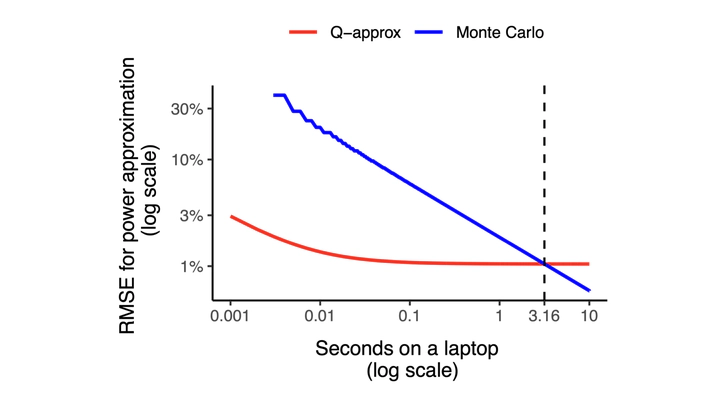
Designing clinical trials requires evaluating multiple operating characteristics (OCs), such as the likelihood of an early stopping decision, the probability of detecting a treatment effect, and the Type I error rate. In most cases, these evaluations are based on computationally intensive Monte Carlo simulations. As the complexity of clinical trials and the use of adaptive designs increase, the computational burden can quickly become prohibitive. We introduce a strategy for rapidly approximating OCs, called the Q-approximation. Our approach is based on quadratic approximations of the log-likelihood and asymptotic arguments. The Q-approximation approach can be applied to any trial design that uses data analysis methods coherent with the likelihood principle, including multistage designs with early stopping, adaptively randomized designs, and designs that leverage external data. We illustrate the approach with several examples and show that it provides an accurate approximation of important OCs while reducing the computation time compared to Monte Carlo simulations. In particular, in our experiments, the standard Monte Carlo approximation of OCs requires 150 to 1,900 times greater computing budget than Q-approximations to achieve comparable levels of accuracy.
Publication
Submitted